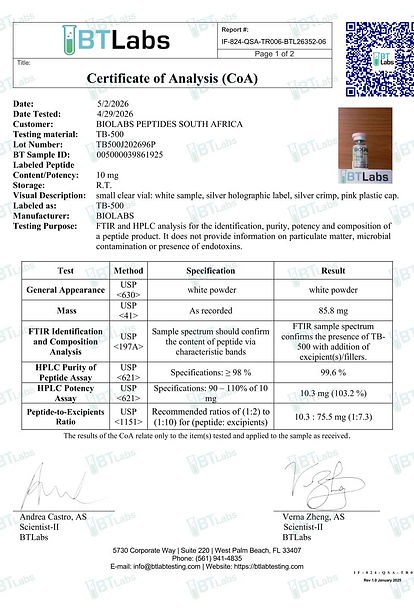
Test Results
BIOLABS is a provider of high purity peptides. Our peptides are upward of 99% purity, available for scientific research and innovation. View our 3rd party testing certificates.
What These Reports Mean
At BioLabs, transparency means independent verification, not marketing claims. The reports you see on this page explain exactly what is inside each vial and why multiple layers of testing matter.
1. Manufacturer vs Independent Testing
All peptides are first tested in-house by the manufacturer. These results are rarely made public because they come from the same source that produces and sells the product.
To remove bias, samples are sent to independent third-party laboratories, primarily Janoshik, widely regarded as the gold standard for peptide analysis.
2. What a Janoshik Report Confirms
A Janoshik report verifies:
-
Identity – the peptide is exactly what it claims to be
-
Purity – typically ≥99%
-
Content – confirms the actual mg per vial
-
Batch linkage – results correspond to a specific production batch
This ensures you are not under-dosed or sold a different compound.
3. Why You May See Multiple Test Sources
One concern in the industry is selective batch testing. To address this, independent groups such as LS Testing who biolabs is apart of purchase peptides directly and test every batch received, regardless of outcome.
This helps confirm batch-to-batch consistency, not just best-case samples.
4. What These Reports Do (and Don’t) Show
These reports confirm purity and accuracy, not full pharmaceutical safety profiles.
Sterility, solvents, and heavy metals are controlled at the manufacturing stage and documented in internal COAs.
Performing full pharmaceutical testing on every retail vial would make products prohibitively expensive.
Why This Matters
Most peptide suppliers provide no public testing at all.
We provide independent verification, clear context, and real data—so researchers know exactly what they are working with.
Our Testing Standards & Transparency
At BioLabs, we believe in full transparency. There is a significant difference between a standard purity report and a full pharmaceutical safety profile, and we want our researchers to understand exactly what they are looking at.
Manufacturing & Sterility
While 3rd-party labs focus on purity, safety is managed at the source. Our manufacturing partners in the EU and Hong Kong operate in professional facilities utilizing:
-
Automated Sterile Filtration: To ensure a contaminant-free environment.
-
Lyophilization: Managed under strict conditions to mitigate microbial and endotoxin risks.
Heavy Metals & Solvents
Screening for heavy metals and residual solvents is managed at the raw material synthesis stage. We rely on the manufacturers' internal COAs (Certificates of Analysis) for these specific screenings.
Note: Performing a full, batch-by-batch residual solvent and heavy metal test on every single retail unit would drive the price up to several times what even "top-tier" clinics charge. We balance high-level testing with affordability.
Why We Do This
In this industry, a Janoshik purity test is the highest level of public transparency available. While 99% of the market provides no data at all, we provide these reports as an extra layer of honesty. We appreciate our customers who understand the science and value knowing exactly what is in their vials.